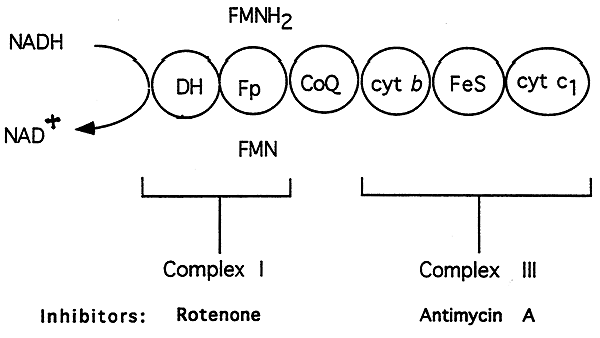
Electron Transport
Respiratory Chain, "Oxidative Phosphorylation"
Purpose of the Pathway: convert NADH and FADH2 into ATP
The principle part of the chain consists of three complexes (I, III, IV) which are integral proteins of the inner mitochondrial membrane (not important to RBC's...) and interact via mobile carriers of electrons.
A fourth complex (II) contains succinate DH.
The components have progressively more positive E0 values leading to electron transfer and energy production for pumping protons out of the matrix.
The components span the inner membrane asymmetrically to facilitate proton pumping.
Complexes I and III:

Complex I, NADH-Q Reductase:
Components: NADH DH, FMN (flavin mononucleotide)
Inhibitor: Rotenone (Rat Poison and Insecticide)
Complex III, Cytochrome Reductase:
Components: b cytochromes, Fe-S-protein center, cytochrome c1
Inhibitor: Antimycin A (Antibiotic)
What Happens:
1. Electrons from NADH enter the chain at NADH-Q Reductase -----> oxidizes NADH
2. NADH donates its electrons to FMN to produce FMNH2 ---------> provides sufficient energy to pump two protons out of the mitochondria.
3. Controlled influx of these protons produces "site one" for ATP production.
4. Electrons are then transfered to coenzyme Q, links complexes I and III, as well as II and III.
5. Coenzyme Q transfers its electrons to cytochrome b and then to a heme Fe-S center (prosthetic group of the cytochromes).
6. Electrons reduce cytochrome c1 with energy yield to pump out two more protons, "site two" for ATP production.
Complexes II and III:
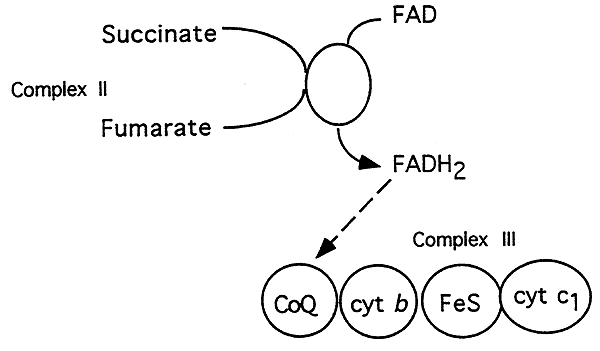
Complex II, Succinate-Coenzyme Q Reductase:
Components: succinate DH, FAD
Inhibitors: none
What Happens:
1. Electron from succinate enter the chain at Succinate-Q Reductase, oxidizes succinate to fumarate (TCA Cycle).
2. Succinate-Q reductase contains FAD, two protons and electrons are accepted from succinate to produce FADH2.
3. Electrons are then transfered to coenzyme Q, which links complexes II and III.
4. Complex II does not produce sufficient energy to pump out protons.
Note: Because electrons passing through complex II bypass complex I, FADH2 yields one fewer ATP molecule than NADH, which feeds electrons through complex I.
Complexes III and IV:
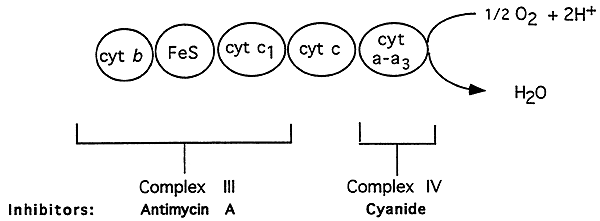
Complex IV, Cytochrome Oxidase:
Components: Cu
Inhibitors: Cyanide
What Happens:
1. Electrons are transfered from cytochrome c1 to cytochrome c.
2. Electrons are then passed to cytochrome a, part of the cytochrome oxidase.
3. Cytochrome a3 accepts electrons from cytochrome a.
4. Cytochrome oxidase interacts directly with molecular O2.
5. Electron transfer through complex IV produces enrgy to release two protons ----->
"site three" for ATP production.
Inhibitors of Electron Transport:
1. Rotenone: inhibits complex I, rat poison and insecticide.
2. Antimycin A: an antibiotic which blocks complex III.
3. Cyanide: inhibits terminal electron transfer to oxygen, Complex IV.
4. Carbon Monoxide: inhibits cytochrome oxidase by competing with an oxygen- binding site, Complex IV.
Asymmetrical Orientation of Electron Transport Chain:
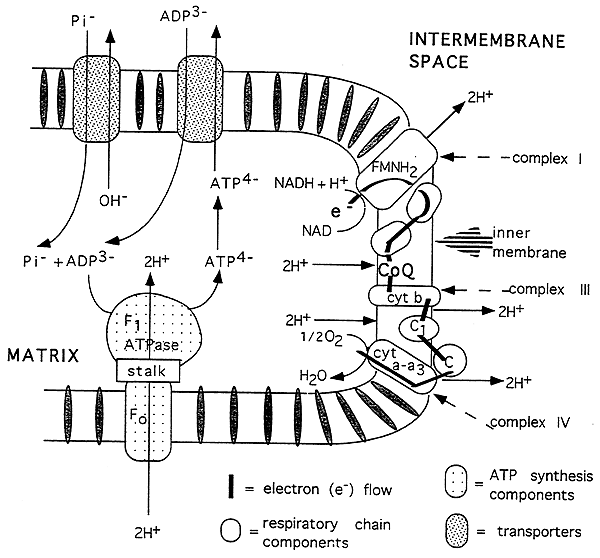
The ATP synthase complex, ADP and phosphate translocases.
Protons are pumped out of the matrix at complexes I, III and IV (2 per complex).
The resulting proton gradient is used to drive ATP synthesis: 2 H+ per ATP produced.
The proton gradient also creates a charge difference across the inner mitochon. membrane.
(membrane potential: more negative on the inside)
ATP Synthase Complex:
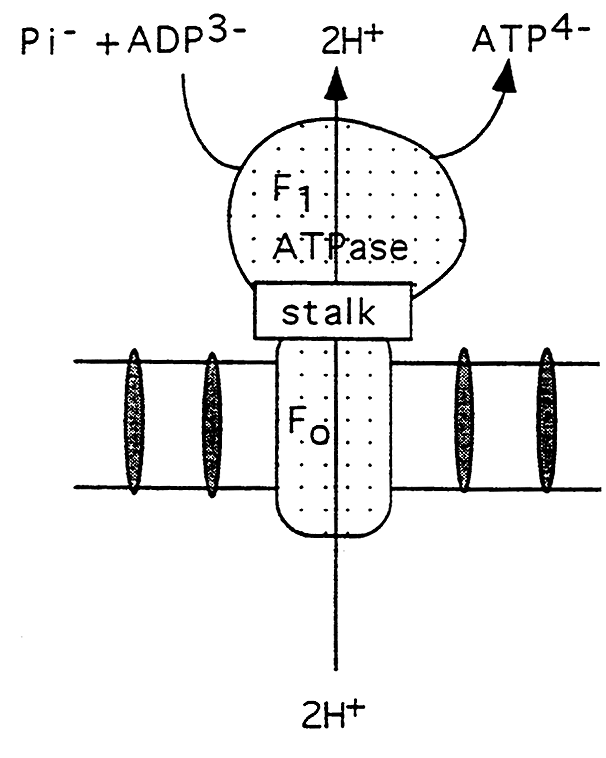
(a) F1 (ATPase) is the sperical protein extending into the matrix.
(b) Contains 5 different subunits and the catalytic site for ATP synthesis.
(c) the complex contains sites that change in their affinity for ATP as protons flow through the complex.
(d) This proton flow allows the ATPase to reverse direction and synthesize ATP.
(e) F0 is the integral oligomer attached to F1 via a stalk.
(f) The F0 contains the proton channel.
(g) The stalk regulates proton flow and ATP synthesis, contains an F1 inhibitor and is the oligomycin sensitive region.
Uncouplers / Respiratory Control / P/O Ratio
Uncouplers: bind protons, are hydrophobic and can dissipate a pH gradient by equilibrating H+ (protons).
i.e. Dinitrophenol (DNP), causes ATP formation to cease but oxygen consumption remains rapid in an attempt by the mitochondria to maintain the proton gradient.
Energy is released as heat and body temperature rises.
Rapid O2 consumption in uncoupling is due to loss of respiratory control.
Respiratory Control:
1. Depends on ADP and Pi regulating O2use.
2. When ATP is high, ADP is limited and O2 use diminishes.
3. O2 use increases as ATP needs rise.
4. With uncoupling and no ATP synthesis, the reduction of O2 vis the respiratory chain is no longer linked to ATP synthesis ------> use is unchecked.
5. The rate of elctron flow is primarily regulated by the ATP:ADP ratio and with uncoupling this control is lost.
6. Because respiration remains rapid the cirtic acid cycle and PDH complex continue at a rapid rate as NADH is maximally oxidized.
Clinical Correlate:
Oligomycin is an antibiotic which binds to the stalk preventing proton flow to the ATPase.
Respiration will diminish because the proton gradient can no longer be utilized.
Respiration will slow even in the presence of large amounts of ADP and phosphate.
(-) citric acid cycle (because of accumulation of NADH)
(-) PDH (increased amounts of NADH and Acetyl CoA)
Effects of ADP, oligomycin and DNP on O2 Consumption:
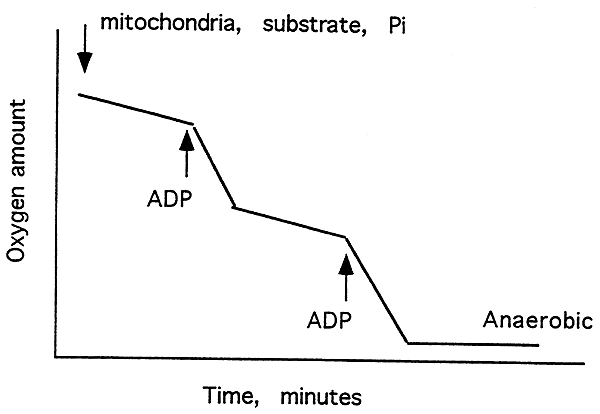
Typical O2recording of mitochondria metabolizing succinate. Repeated additions of ADP increase O2 consumption until either ADP or O2 is depleted. This increased consumption reflects respiratory control.
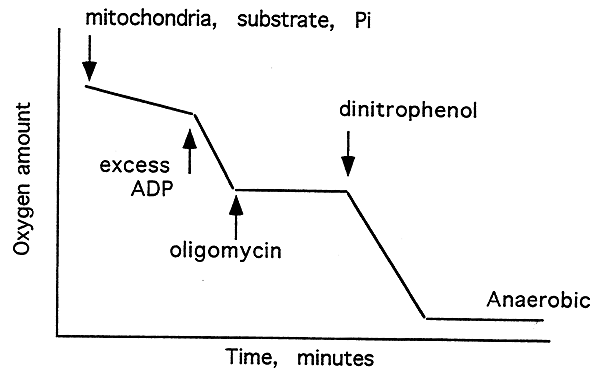
If an XS of ADP is added, followed by oligomycin, then O2 consumption ceases. if uncoupler is now added the rate of repiration (O2 consumption) increases dramatically as the proton gradient collapses as quickly as it forms.
P / O Ratio:
The P / O (phosphate:oxygen) ratio represents the amount of Pi used (ATP synthesized) per atom of oxygen consumed.
1. Electron flow from NADH to O2 pumps protons ar three sites to yield 3 ATP (P / O = 3)
2. Succinate (via FADH2) bypasses site 1 giving 2 ATP (P / O = 2)
3. Uncoupling: P / O = 0, oxygen is consumed with no ATP production.
Summary Diagram:
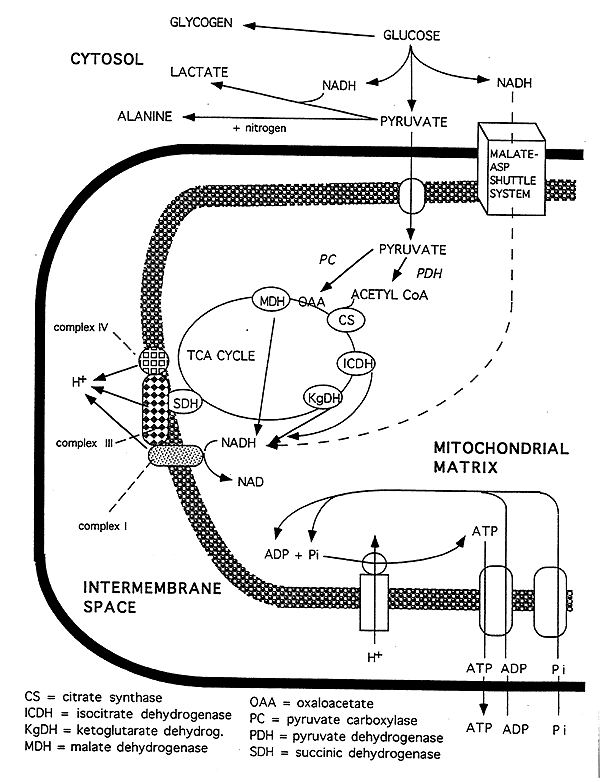
Malate-Aspartate Shuttle: NADH enters at Complex I producing 3 ATP.
Glycerol Phosphate Shuttle: converts NADH to FADH2 in the cytosol then the FADH2 enters the chain at Complex II producing 2 ATP.
© Dr. Noel Sturm 2014